1. Temperature of the Oceans.
The temperature of seawater is fixed at the surface by its heat exchange with the atmosphere. At the equator, the energy received from the sun is 4x times greater than at the poles. There is more heat to the earth's surface in the tropical regions- this is why the oceans surface are the warmest there. Heat is transferred from the low latitudes (equator) to the high latitudes (poles) by wind and ocean currents.
The oceans have a high specific heat capacity, so that seasonal variations in temperature only vary by a few degrees, except in very shallow water. Land has a higher variation diurnally (daily) and seasonally.
It takes more energy to heat up the water than it does to heat up the land. However, it takes water longer to lose heat. This is why land
The oceans have a high specific heat capacity, so that seasonal variations in temperature only vary by a few degrees, except in very shallow water. Land has a higher variation diurnally (daily) and seasonally.
It takes more energy to heat up the water than it does to heat up the land. However, it takes water longer to lose heat. This is why land
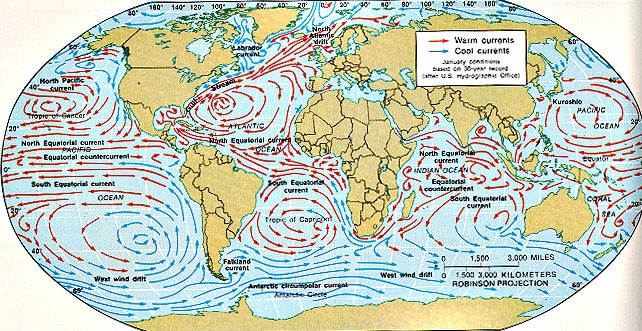
2. The distribution of the oceans and ocean currents.
The atmosphere and the oceans link together and regulate global climatic conditions. Warm ocean currents move water away from the equator, whereas cold ocean currents move water away from the cold regions towards the equator. The major currents move huge masses of water over long distances- the warm Gulf Stream transports 55 million cubic metres per second. Without it, temperate lands of northwestern Europe would be more like the sub-Arctic. The cold Peru Current and the Benguela Current of SW Africa bring in nutrient rich waters dragged to the surface by offshore winds.
The Great Ocean Conveyor Belt
- In addition to the transfer of energy by wind, and ocean currents, there is also the transfer of energy by deep sea ocean currents. Oceanic Convection occurs from the polar regions, where cold, salty water sinks into the depths and makes its way towards the equator.
- The densest water is found in the deep Antarctic waters- here seawater freezes to form ice at a temperature of around about -2C. It sweeps around Antarctica at a depth of around 4km.
- It then spreads into the deep basins of the Atlantic, Pacific and Indian oceans.
- Surface currents bring warm water to the North Atlantic from the Indian and Pacific. These waters then give up their heat to cold winds which blow over Canada across the North Atlantic. The water then sinks and starts the reverse convection of the deep ocean current. The amount of heat given up is about 1/3 of the energy received from the sun.
- This means that the North Atlantic is warmer than the North Pacific, so there is more evaporation there although this is proportional.
3. Density and Salinity of the Oceans

Salinity
There are many chemicals in seawater that make it salty. Most of them get there from rivers carrying chemicals dissolved out of rock and soil. The main one is sodium chloride, often just called salt. Most seawater has about 35g (7 teaspoons) of salt in every 1000g (about a litre) of water. This doesn’t sound very much, but it would take three 6m shipping containers full of salt to make an Olympic-size swimming pool as salty as the sea.
The commonest way to record salinity is to measure the amount of salt in 1000g of water, so it is referred to as ‘parts per thousand’ or ppt. Most of the ocean has a salinity of between 34ppt and 36ppt.
Some properties of water are changed by having salt in it:
- Salt makes seawater more dense than freshwater.
- Salty water needs to be colder than freshwater before it freezes.
The salinity of the ocean varies from place to place, especially at the surface. Much of the ocean has salinity between 34ppt and 36ppt, but there are places that tend to be higher or lower.
Places of higher salinity
Much of the open ocean has a salinity between 34ppt and 36ppt. Salinity is controlled by a balance between water removed by evaporation and freshwater added by rivers and rain.
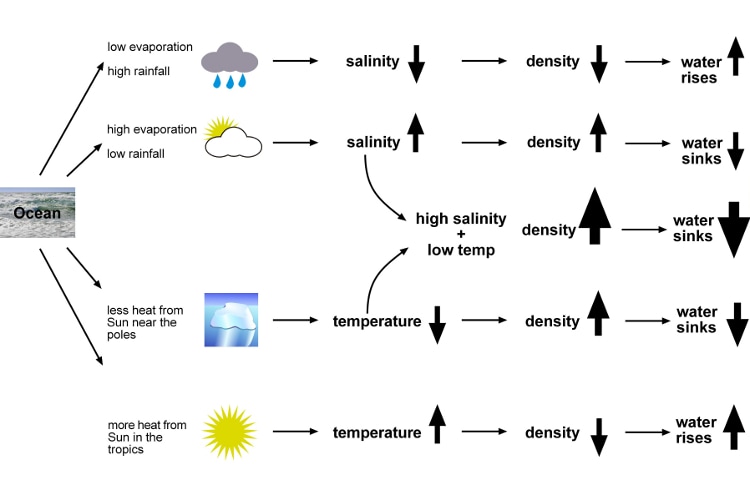
There are parts of the ocean where hardly any rain falls but warm dry winds cause lots of evaporation. This evaporation removes water – when water vapour rises into the atmosphere, it leaves the salt behind, so the salinity of the seawater increases. This causes the seawater to become denser. You can see on the map that the north and south Atlantic have high salinity – these are areas where there are strong winds and not much rain.
The Mediterranean Sea in Europe has very high salinity – 38ppt or more. It is almost closed from the main ocean, and there is more evaporation than there is rain or extra freshwater added from rivers.
Places of lower salinity
Some parts of the ocean have lots of rain. The freshwater added at the surface dilutes the seawater, reduces the salinity and so makes the seawater less dense. Seawater can also be less saline near land, where rivers add freshwater.
The ocean around Antarctica has a low salinity of just below 34ppt, and around the Arctic it is down to 30ppt in places. Thawing icebergs add freshwater – icebergs that have broken off ice sheets formed over land do not contain salt, and the freezing of seawater into ice floes removes more salt.
Density
The density of fresh water and seawaterFresh water and seawater have very different physical properties. Imagine a freshwater lake in winter. As the air temperature falls the temperature of the water at the surface decreases and its density changes.
The density of seawater plays a vital role in causing ocean currents and circulating heat because of the fact that dense water sinks below less dense. Salinitytemperature and depth all affect the density of seawater.
Density is a measure of how tightly a certain amount of matter is packed into a given volume. The more the stuff is packed in, the higher the density. Density can be calculated by dividing an object’s mass by its volume. It is commonly measured in grams per millilitre or grams per cubic centimetre.
Seawater is not just water – it has lots of chemicals packed into it. This means it is denser than pure water. The higher the salinity, the higher the density.
Seawater density varies from place to place because it is affected by salinity and temperature. This means that ships float higher or lower in the water, depending on the density of the ocean. If you look near the waterline of a cargo ship, you should find the International Load Line, once called the Plimsoll Line. This shows the limit of where the fully loaded ship should sit in waters of different densities.
High salinity makes water denser. This is because there is more salt packed into the water.
High temperature makes water less dense. As water gets warmer, its molecules spread out, so it becomes less dense. As it gets colder, it becomes denser. Most chemicals get denser when they turn from a liquid to a solid, but water is different. When liquid water freezes into solid ice, it becomes less dense. When ice forms, water molecules arrange themselves into a rigid but open pattern. This structure is less dense than the liquid water, so ice floats.
Deep water is denser than shallow water. The water molecules are packed together more tightly because of the weight of water above pushing down.
Dense water sinks below less dense water. This is the principle that drives the deep ocean currents that circulate around the world. A combination of high salinity and low temperature near the surface makes seawater dense enough to sink into the deep ocean and flow along the bottom of the basins.