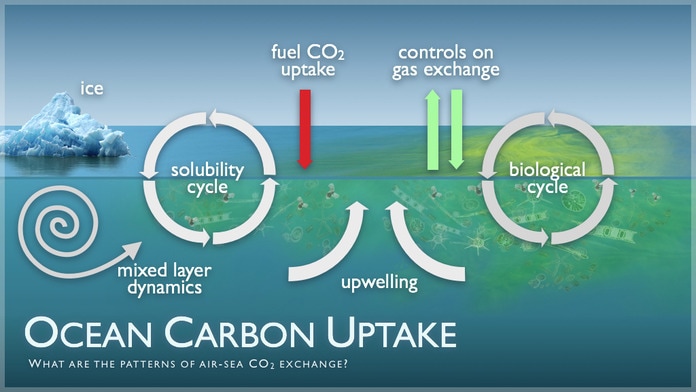
1. The carbon cycle

A summary of how CO2 works as a source and a store within the carbon cycle.
•Direct transfer between ocean and atmosphere and vice versa.
•Photosynthesis by plankton absorbs CO2.
•Some CO2 passes through the food chain.
•Death of fish = decomposition = CO2 in sediment.
•Fossilisation and some sediment is destroyed at subduction zones.
•CO2 released by volcanic activity.
•Direct transfer between ocean and atmosphere and vice versa.
•Photosynthesis by plankton absorbs CO2.
•Some CO2 passes through the food chain.
•Death of fish = decomposition = CO2 in sediment.
•Fossilisation and some sediment is destroyed at subduction zones.
•CO2 released by volcanic activity.
2. Stores and sources of carbon
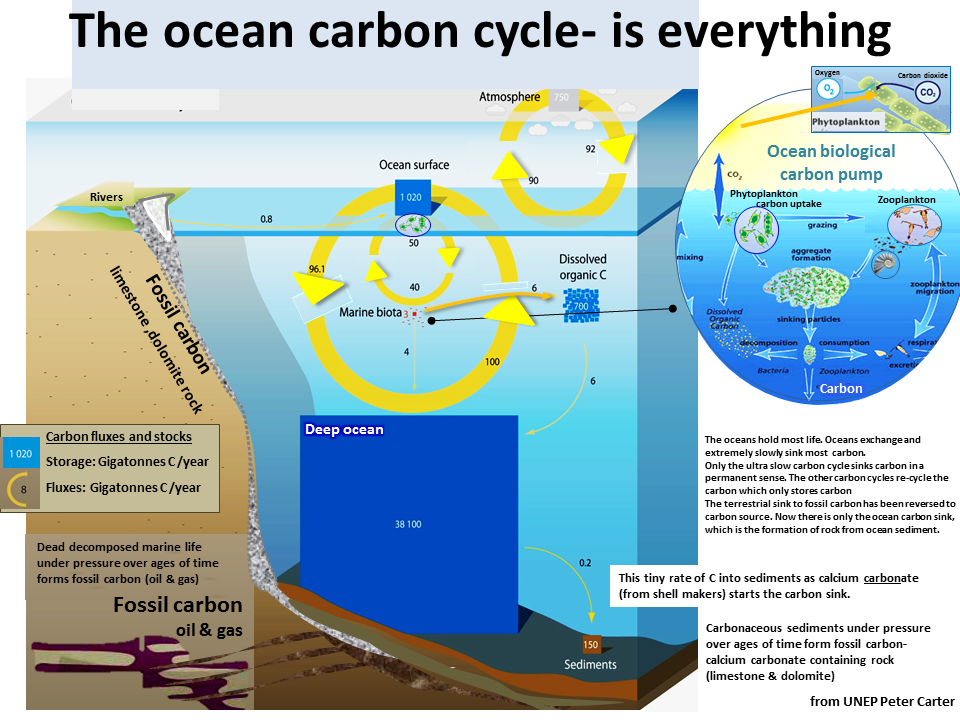
Carbon moves around the earth between the atmosphere and oceans and between the atmosphere and the biosphere. The oceans hold the most carbon dioxide. Some of it is stored in the oceans, and sometimes the oceans act as a source. The oceans contain about 50 times more CO 2 than the atmosphere and 19 times more than the land biosphere. CO 2 moves between the atmosphere and the ocean by molecular diffusion when there is a difference between CO 2 gas pressure (pCO 2 ) between the atmosphere and oceans. For example, when the atmospheric pCO 2 is higher than the surface ocean, CO 2 diffuses across the air-sea boundary into the sea water.
The oceans are able to hold much more carbon than the atmosphere because most of the CO 2 that diffuses into the oceans reacts with the water to form carbonic acid and its dissociation products, bicarbonate and carbonate ions . The conversion of CO 2 gas into nongaseous forms such as carbonic acid and bicarbonate and carbonate ions effectively reduces the CO 2 gas pressure in the water, thereby allowing more diffusion from the atmosphere.
The oceans are mixed much more slowly than the atmosphere, so there are large horizontal and vertical changes in CO 2 concentration. In general, tropical waters release CO 2 to the atmosphere, whereas high-latitude oceans take up CO 2 from the atmosphere. CO 2 is also about 10 percent higher in the deep ocean than at the surface. The two basic mechanisms that control the distribution of carbon in the oceans are referred to as the solubility pump and the biological pump.
Before the industrial revolution, atmospheric concentrations were at a constant because carbon being removed from the oceans.
The oceans can store CO2 by absorbing it in two main ways: physically- the CO2 is dissolved. This is part of the solubility pump. CO2 dissolves into the cold water near the poles (the water here isn't directly and consistently heated by insolation), as the currents sink into the deep ocean, they take the CO2 with them. Over time, the water returns to the surface and the ocean emits CO2 into the atmosphere, within the Tropics. This system is quite variable because mixing is a slow and uneven process this means that there are different amounts of CO2 in different parts of the ocean. Deep or shallow determines how much is stored, tropical or polar will affect the amount of dispersion and how much becomes a source.
The oceans also store CO2 through phytoplankton which takes up CO2, zooplankton eats the phytoplankton, and this continues up through the food web. This is the biological pump.
How does anthropogenic climate change affect this?
Today, CO 2 concentrations in the atmosphere are increasing as a direct result of human activities such as deforestation and the burning of fossil fuels (e.g., coal and oil). Over the past 150 years, CO 2 concentrations in the atmosphere have increased by as much as 30 percent (from 280 to 370 ppm).
All trees, nearly all plants from cold climates, and most agricultural crops respond to increasing atmospheric CO 2 levels by increasing the amount of CO 2 they take up for photosynthesis . It is believed that the increased uptake in land plants from rising atmospheric CO 2 levels roughly counterbalanced the CO 2 released from cutting down tropical rain forests and other agricultural practices in the decade of the 1980s. In the 1990s, the land biosphere was estimated to take up approximately 1 Pg more CO 2 than it released each year.
Most of the CO 2 released from the burning of fossil fuels and other human activities (e.g., cement manufacturing) is stored either in the atmosphere or in the oceans. The CO 2 that remains in the atmosphere acts as a greenhouse gas, absorbing long-wavelength radiation (heat) in the atmosphere. CO 2 taken up by the oceans does not affect the Earth's heat balance, so an understanding of the air-sea exchange of CO 2 is an essential part of understanding the Earth's climate system and the potential impact of future CO 2 emissions.
Read more: http://www.waterencyclopedia.com/Bi-Ca/Carbon-Dioxide-in-the-Ocean-and-Atmosphere.html#ixzz4d4TA3mSW
Today, CO 2 concentrations in the atmosphere are increasing as a direct result of human activities such as deforestation and the burning of fossil fuels (e.g., coal and oil). Over the past 150 years, CO 2 concentrations in the atmosphere have increased by as much as 30 percent (from 280 to 370 ppm).
All trees, nearly all plants from cold climates, and most agricultural crops respond to increasing atmospheric CO 2 levels by increasing the amount of CO 2 they take up for photosynthesis . It is believed that the increased uptake in land plants from rising atmospheric CO 2 levels roughly counterbalanced the CO 2 released from cutting down tropical rain forests and other agricultural practices in the decade of the 1980s. In the 1990s, the land biosphere was estimated to take up approximately 1 Pg more CO 2 than it released each year.
Most of the CO 2 released from the burning of fossil fuels and other human activities (e.g., cement manufacturing) is stored either in the atmosphere or in the oceans. The CO 2 that remains in the atmosphere acts as a greenhouse gas, absorbing long-wavelength radiation (heat) in the atmosphere. CO 2 taken up by the oceans does not affect the Earth's heat balance, so an understanding of the air-sea exchange of CO 2 is an essential part of understanding the Earth's climate system and the potential impact of future CO 2 emissions.
Read more: http://www.waterencyclopedia.com/Bi-Ca/Carbon-Dioxide-in-the-Ocean-and-Atmosphere.html#ixzz4d4TA3mSW
3. Impacts and feedback mechanisms
 Source: http://news.bbc.co.uk/1/hi/sci/tech/7933589.stm
Source: http://news.bbc.co.uk/1/hi/sci/tech/7933589.stm
Acidification
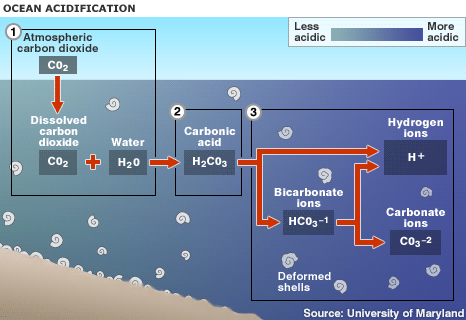
1. Up to one half of the carbon dioxide (CO2) released by burning fossil fuels over the past 200 years has been absorbed by the world's oceans.
2. Absorbed CO2 in seawater (H2O) forms carbonic acid (H2CO3), lowering the water's pH level and making it more acidic
3.This raises the hydrogen ion concentration in the water, and limits organisms' access to carbonate ions, which are needed to form hard parts
The oceans have absorbed up to half of this excess CO2, which has resulted in changes in the chemistry of surface seawater. The CO2 in the water, which leads to the formation of carbonic acid, has caused the pH of surface oceans to fall by 0.1 units, and it is projected to fall a further 0.3-0.4 pH units by the end of the century. The shift in the waters' chemical make-up not only increases its acidity, but reduces the availability of carbonate ions, which many creatures use to build shells and skeletons out of calcium carbonate.
The decrease in available carbonate ions means that organisms, such as plankton, coral and molluscs, struggle to build or maintain their protective or supportive structures.
Researchers believe that areas with relatively low pH, such as the eastern Pacific, could be the result of the upwelling of deeper, colder, CO2-richer waters. However, no region is expected to escape the impact of falling pH. As a result, marine biologists say that a number of species and ecosystems face an uncertain future:
Warm-water coral reefs
Recipe for rescuing reefsEvidence suggests that the calcification rates of these corals will be reduced by up to 60%, say US researchers writing in the journal Current Biology. They say a reduction of this magnitude could adversely affect reef structures, as growth depends on corals' ability to build faster than the skeleton is eroded. Weaker structures are likely to be prone to greater degrees of erosion from storms and heavy wave action.
Cold-water corals
Found throughout the world's oceans, cold-water corals can provide vital habitat for several commercially important fish species. Forecasts suggest that about 70% of the corals could find themselves under threat by the end of the century.
Plankton
These tiny organisms play an important role in the marine food chain. Several groups of plankton produce calcium carbonate, and could see their distribution curtailed by ocean acidification. Scientists agree that more research is required in order to better understand the impact of oceans' falling pH on the tiny creatures.
Some species, such as coccolithophores (single-celled algae), have shown a marked decrease in calcification rates when exposed to CO2-rich water.
However, others species were largely unaffected.
Invertebrates
Some molluscs, including mussels and oysters, are expected to be adversely affected by ocean acidification.
As with other creatures, the main impact is expected to be thin or deformed shells.
Juveniles have been shown to be more susceptible than adults to limited carbonate ions, which could have long-term consequences as far as viable populations are concerned.
But not all habitats suffer as a result of ocean acidification. For example, sea grasses grow better in CO2-rich waters.
The grass offers a valuable feeding and spawning site for a variety of species, including a number of commercially valuable fish.
However, more research is needed to see if the local benefits from the sea grasses are not outweighed by the wider disruption to the marine food chain.
Feedback mechanisms:
These natural "carbon pumps" are showing signs of being disrupted, say researchers. For example, ocean acidification could reduce plankton blooms, resulting in less CO2 being absorbed from the atmosphere. In theory, the extra CO2 in the atmosphere could lead to an acceleration in global warming, which will warm the oceans. As a result, the warmer waters will not be able to absorb as much carbon dioxide as cooler seas. So even less CO2 is taken from the atmosphere, resulting in more of the greenhouse gas being available to warm the planet. The potential of these "feedback" mechanisms to disrupt the planet's climate system is one of the reasons why marine scientists are calling for urgent action to be taken to stabilise, and eventually reduce, emissions. (Source: BBC)
1. Up to one half of the carbon dioxide (CO2) released by burning fossil fuels over the past 200 years has been absorbed by the world's oceans.
2. Absorbed CO2 in seawater (H2O) forms carbonic acid (H2CO3), lowering the water's pH level and making it more acidic
3.This raises the hydrogen ion concentration in the water, and limits organisms' access to carbonate ions, which are needed to form hard parts
The oceans have absorbed up to half of this excess CO2, which has resulted in changes in the chemistry of surface seawater. The CO2 in the water, which leads to the formation of carbonic acid, has caused the pH of surface oceans to fall by 0.1 units, and it is projected to fall a further 0.3-0.4 pH units by the end of the century. The shift in the waters' chemical make-up not only increases its acidity, but reduces the availability of carbonate ions, which many creatures use to build shells and skeletons out of calcium carbonate.
The decrease in available carbonate ions means that organisms, such as plankton, coral and molluscs, struggle to build or maintain their protective or supportive structures.
Researchers believe that areas with relatively low pH, such as the eastern Pacific, could be the result of the upwelling of deeper, colder, CO2-richer waters. However, no region is expected to escape the impact of falling pH. As a result, marine biologists say that a number of species and ecosystems face an uncertain future:
Warm-water coral reefs
Recipe for rescuing reefsEvidence suggests that the calcification rates of these corals will be reduced by up to 60%, say US researchers writing in the journal Current Biology. They say a reduction of this magnitude could adversely affect reef structures, as growth depends on corals' ability to build faster than the skeleton is eroded. Weaker structures are likely to be prone to greater degrees of erosion from storms and heavy wave action.
Cold-water corals
Found throughout the world's oceans, cold-water corals can provide vital habitat for several commercially important fish species. Forecasts suggest that about 70% of the corals could find themselves under threat by the end of the century.
Plankton
These tiny organisms play an important role in the marine food chain. Several groups of plankton produce calcium carbonate, and could see their distribution curtailed by ocean acidification. Scientists agree that more research is required in order to better understand the impact of oceans' falling pH on the tiny creatures.
Some species, such as coccolithophores (single-celled algae), have shown a marked decrease in calcification rates when exposed to CO2-rich water.
However, others species were largely unaffected.
Invertebrates
Some molluscs, including mussels and oysters, are expected to be adversely affected by ocean acidification.
As with other creatures, the main impact is expected to be thin or deformed shells.
Juveniles have been shown to be more susceptible than adults to limited carbonate ions, which could have long-term consequences as far as viable populations are concerned.
But not all habitats suffer as a result of ocean acidification. For example, sea grasses grow better in CO2-rich waters.
The grass offers a valuable feeding and spawning site for a variety of species, including a number of commercially valuable fish.
However, more research is needed to see if the local benefits from the sea grasses are not outweighed by the wider disruption to the marine food chain.
Feedback mechanisms:
These natural "carbon pumps" are showing signs of being disrupted, say researchers. For example, ocean acidification could reduce plankton blooms, resulting in less CO2 being absorbed from the atmosphere. In theory, the extra CO2 in the atmosphere could lead to an acceleration in global warming, which will warm the oceans. As a result, the warmer waters will not be able to absorb as much carbon dioxide as cooler seas. So even less CO2 is taken from the atmosphere, resulting in more of the greenhouse gas being available to warm the planet. The potential of these "feedback" mechanisms to disrupt the planet's climate system is one of the reasons why marine scientists are calling for urgent action to be taken to stabilise, and eventually reduce, emissions. (Source: BBC)
4. Essay practice
How would you write an essay about this?
May 2013: Examine the role of oceans as a store and source of carbon dioxide. (10 marks)
This is a complicated essay to tackle because you need to be confident about the two aspects. It is easier to write about the oceans as a store than a source. I would approach it in the following way (remember that this is not prescriptive but a suggestion).
Introduction
In the essay, you need to decide which aspect is most important because the command word is "examine". The other important factor in this question is timescales. In the past, the source element was important but today, the store element is important because we already have enough in our oceans.
Argument 1: The oceans as a source
Argument 2: The ocean as a store
This is where we feel confident because we learn about this in several subjects. There are ways of being balanced even if we says lightly more about this.
The oceans store carbon in the following ways:
The discussion
This is where you would summarise your main points and make a clear decision- don't hedge too much and ensure that you answer the question. For example:
The oceans have been critical in Earths atmospheric development and have provided an essential source of carbon. This process take millions of years though, so while being very important over a long time scale, since the industrial revolution the role as a store has become essential. In the modern world, it is the oceans ability to store carbon which is so critical to managing and maintaining the delicate balance between atmosphere and ocean and anthropogenic climate change. If the oceans role as a store begins to change, this could have devastating impacts on the Earths carbon cycle and balance.
Or similar.
Now remember, this question has already appeared in 2013, so if carbon comes up again, it will likely have a different focus, so you should make sure you are clear about the links carbon has to other parts of this topic- notice how i have linked it to corals (as above)
May 2013: Examine the role of oceans as a store and source of carbon dioxide. (10 marks)
This is a complicated essay to tackle because you need to be confident about the two aspects. It is easier to write about the oceans as a store than a source. I would approach it in the following way (remember that this is not prescriptive but a suggestion).
Introduction
- Where does carbon come from naturally? - This is where you can introduce a bit of background on the carbon cycle.
- Why is it important to our planet? - You could briefly introduce the greenhouse effect here but don't spend too long.
- Briefly outline that carbon the oceans are important as a source and store of carbon.
In the essay, you need to decide which aspect is most important because the command word is "examine". The other important factor in this question is timescales. In the past, the source element was important but today, the store element is important because we already have enough in our oceans.
Argument 1: The oceans as a source
- In Earths long history a primary source of carbon was volcanic activity, but this is less important today.
- Over Earths history many organisms have taken on carbon, when the organisms die they are deposited on the sea floor. It moves up the food chain from phytoplankton. Phytoplankton photosynthesises creating organic carbon dioxide. This is then eaten by the next organism. Over millions of years the organisms and so on.
- The sea floor is then subjected at destructive plate margins and then when a volcano erupts it is emitted back into the atmosphere. This comes from the ocean floor so is a source of carbon.
- Deep oceans contain 38000GT of carbon. It got here because of the deep ocean currents- the thermocline conveyor belt. This happens over a long period- somewhere around 800 years!
- There is also some transfer between the surface and the atmosphere.
Argument 2: The ocean as a store
This is where we feel confident because we learn about this in several subjects. There are ways of being balanced even if we says lightly more about this.
The oceans store carbon in the following ways:
- The oceans absorb carbon in two ways- physically and biologically:
- CO2 dissolves into cold ocean water near the poles, and it is carried to the deep ocean by sinking currents. Over time, the water returns to the surface and the ocean emits CO2 into the atmosphere in tropical regions.
- Another process that moves CO 2 away from the surface ocean is called the biological pump. Growth of marine plants (e.g., phytoplankton) takes CO 2 and other chemicals from sea water to make plant tissue. Microscopic marine animals, called zooplankton, eat the phytoplankton and provide the basis for the food web for all animal life in the sea. Because photosynthesis requires light, phytoplankton only grow in the nearsurface ocean, where sufficient light can penetrate. Although most of the CO 2 taken up by phytoplankton is recycled near the surface, a substantial fraction, perhaps 30 percent, sinks into the deeper waters before being converted back into CO 2 by marine bacteria. Only about 0.1 percent reaches the seafloor to be buried in the sediments.
- It can be stored in coral reefs which are carbon sinks.
The discussion
- The system was previously in balance.
- The process of volcanism absorbing carbon and repurposing it through sediment and sea floor spreading until it is subjected and reemitted into the atmosphere takes millions of years. So could potentially keep a lot of carbon out of the atmosphere, except that this is no longer the biggest way of processing the carbon, and is in fact now relatively minor.
- Today the roles have been muddied by anthropogenic climate change. As we burn more fossil fuels, the oceans should be able to absorb more carbon. And they have absorbed up to 50% of the excess carbon created. They are able to store more because they absorb the CO2, and the mixing with H2O means that its stored as carbonic acid.
- But, this has changed the natural cycle as the sea waters PH level has changed. This has led to the acidity of the oceans increasing which in turn has led to a reduction in available carbonate ions, which is essential for shell and coral building, leading to less carbon being stored biologically.
- There is strong evidence to suggest that if we keep putting more CO2 into the atmosphere, and the climate continues to warm less CO2 will be able to be absorbed which means that the importance of the oceans as a store cannot be overstated. Currently, they absorb and remove carbon to the deep ocean, they process the carbon into carbonate for animal shells, and provide a source of carbon for the creation of coral reefs, an important carbon sink, which man is killing through poor practice and management and some natural processes relating to climate change.
This is where you would summarise your main points and make a clear decision- don't hedge too much and ensure that you answer the question. For example:
The oceans have been critical in Earths atmospheric development and have provided an essential source of carbon. This process take millions of years though, so while being very important over a long time scale, since the industrial revolution the role as a store has become essential. In the modern world, it is the oceans ability to store carbon which is so critical to managing and maintaining the delicate balance between atmosphere and ocean and anthropogenic climate change. If the oceans role as a store begins to change, this could have devastating impacts on the Earths carbon cycle and balance.
Or similar.
Now remember, this question has already appeared in 2013, so if carbon comes up again, it will likely have a different focus, so you should make sure you are clear about the links carbon has to other parts of this topic- notice how i have linked it to corals (as above)